| Главная » Файлы » Иностранные языки » Английский язык |
| 22.12.2013, 21:18 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
INTRODUCTION RIGIDITY of WATER Scum and adjournment of salts on home appliances (for example, in teapots), film on tea etc. - all this parameters of rigid water. Use of such water for the economic purposes causes a number of inconveniences. For example, the charge of a soap is increased at washing, slowly boil soft meat and vegetables, service life of home appliances decreases. Interrelation of rigidity of water and education of stones in kidneys now is known. The rigidity of drinking water under the working standards should be not higher 7 mg-ecv/g, and only in the special cases it is supposed up to 10 mg-ecv/g. For the industrial purposes use of rigid water is inadmissible. The general rigidity of water is a set of properties caused by the contents in it ions of magnesium and calcium. Hard water must be soften before use. The first way – reagent method i.e., addition slaked to exhaust and soda Na2CO3 (a limy way), addition of polyphosphates. The second way - application of cationits , i.e., synthetic ion-exchange pitches (filtering). Ion-exchange pitches These are substances capable to an ionic exchange at contact to solutions of electrolits. Ionic-exchange clearing allows to take and utilize a wide spectrum of polluting substances: heavy metals, chrom, nitrates and nitrites, cyanic connections, radioactive substances, and also умягчает and unironing water. Thus the high degree of clearing (up to a level of maximum concentration limit) is reached. Except for that ionits are used for unsalting waters during water-preparation. Inorganic and organic ionits can be natural (for example: zeolites, cellulose, peat, wood) and synthetic (silica gel and the most important ion-exchange pitches). Depending on a degree dissociation of ion-exchange pitches can be strong and weak. Depending on a sort of ions which are connected to active groups of ionits, distinguish the following its form: for cationits - the hydrogen form (H-form) and the salt form, when active groups are connected to ions of metals (for example, Na-form, NH4-form), for anionits OH-form, Cl-form, etc. Ability of ionits to a full exchange is characterized by exchange capacity which is equal to number of its active groups participating in an exchange. For the quantitative characteristic of ion-exchange properties of ionits usually define their dynamic and sometimes full (general) exchange capacity (static). Main requirements to ionits, used for water treating: high exchange capacity, high speed of an ionic exchange, sufficient stability in relation to acids, alkalis, oxidizers and reducers, insolubility in water, organic solvents and solutions of electrolits and limited swelling capacity. In water-preparation in a conditions of life are frequently used high-acid cationits domestic and import manufacturers mainly for softening and iron removal of waters. An example: the structure of cationits can be expressed formula Na2R, where Na+ - rather mobile cation. If to pass hard water through layers of cationits ions of sodium exchange on ions of calcium and magnesium: Ca 2 + + Na2R = 2Na + + CaR Mg 2 + + Na2R = 2Na + + MgR Thus ions Ca2+ and Mg2+ pass from a solution to cationit, and ions Na+-from cationit in a solution, and rigidity is eliminated. After pauperization of cationit ions of Na+ cationits usually recycle. Them maintain in solution NaCl where there is a return replacement - ions Na+ pass in cationit, and ions Ca2+ and Mg2+ - in a solution: CaR + 2Na+ = Na2R + Ca 2+ MgR + 2Na + = Na2R + Mg 2+ After that regenerated cationit can be used for mitigation of new portions of hard water. The degree of regeneration is influenced with type of ionits, structure of the sated layer, the nature, concentration and a solution of recycling substance, temperature, time of contact and the charge of reagents. Restoration of exchange capacity at regeneration usually makes 60 - 100 %. Kinds of rigidity General rigidity. It is defined by total concentration of ions of calcium and magnesium. Represents the sum of carbonate (time) and uncarbonate (constant) rigidity. Carbonate rigidity. It is caused by presence in water of hydrocarbonates and carbonates (at рН> 8.3) calcium and magnesium. The given type of rigidity almost completely is eliminated at boiling waters and consequently refers to as time rigidity. At heating water hydrocarbonates break up with formation of a coal acid and settling out of a carbonate of calcium and oxyhydroxide of magnesium. Origin of rigidity Ions of calcium (Ca2+) and magnesium (Mg2+), and also others alkali-ground metals causing rigidity, are present at all mineralizeed waters. Their source are natural deposits of limestones, plaster and dolomite. Ions of calcium and magnesium act in water as a result of interaction dissolved dioxide of carbon with minerals and at other processes of dissolution and chemical aeration of rocks. As a source of these ions, the microbiological processes proceeding in soil on the area of a reservoir can serve in ground adjournment, and also sewage of the various enterprises also. Rigidity of water changes over a wide range and there is a set of types of classifications of water on a degree of its rigidity. Usually in little mineralizeed waters prevails (up to 70%-80 %) the rigidity caused by ions of calcium (though in separate rare cases magnesian rigidity can reach(achieve) 50-60 %). With increase in a degree of a mineralization of water the contents of ions of calcium (Са2+) quickly falls and seldom exceeds 1 g/l. The contents of ions of magnesium (Mg2+) in high mineralizeed waters can reach several grammes, and in salty lakes - tens grammes on one litre of water.As a whole, rigidity of superficial waters, as a rule, is less than rigidity of waters underground. Rigidity of superficial waters is subject to appreciable seasonal fluctuations, reaching usually the greatest value at the end of winter and the least during a high water when it is plentifully diluted soft rain and thawed snow. Sea and ocean water have very high rigidity (tens and hundreds in mg - ecv/l) Influence of rigidity From the point of view of application of water for drinking needs, its acceptability on a degree of rigidity can vary essentially depending on local conditions. The threshold of taste for an ion of calcium lays (in recalculation on a mg - equivalent) in a range of 2-6 mg - ecv/l, depending on corresponding of anionits, and a threshold of taste for magnesium and than that is lower. In some cases water is comprehensible to consumers with rigidity above 10 mg-ecv/l. High rigidity worsens organoleptic properties of water, giving to it bitterish taste and having negative an effect on bodies of digestion. The world Organization of Public health services (CART) does not offer any recommended size of rigidity under indications of influence on health. In materials the CART is spoken that though a number of researches and has revealed statistically inverse relationship between rigidity of potable water and the cardiovascular diseases, the available data are not sufficient for a conclusion about causal character of this communication. Similarly, unequivocally it is not proved, that soft water renders a negative effect on balance of mineral substances of an organism of the person At the same time, depending on рН and alkalinity, water with rigidity of 4 mg-ecv/l can above cause in distributive system adjournment of slags and scums (a carbonate of calcium), is especial at heating. For this reason norms of Boiler inspection enter very rigid requirements to size of rigidity of the water used for a meal of boilers (0.05-0.1 mg-ecv/l). Besides at interaction of salts of rigidity with washing substances (the soap, detergent powders, shampoos) occurs formation of "soap slags" as foam. It results not only in the significant overexpenditure of washing-up liquids. Such foam after drying remains as a strike on the sanitary technician, linen, a human skin, on hair (unpleasant feeling of "rigid" hair well-known to much). The main negative influence of these slags on the person is that they destroy a natural fatty film with which the normal skin is always covered and hammer its time. An attribute of such negative influence is characteristic "scratch" of cleanly washed up skin or hair. It appears, that the irritation causing in some people feeling "soapiness" after using soft water is an attribute of that the protective fatty film on a skin is whole and safe. It also slides. At the same time, it is necessary to mention and other side of a medal. Soft water with rigidity less than 2 mg-ecv/l has low buffer capacity (alcalinity) and can, depending on a level рН and of some other factors, render the increasedcorrosive attack to water pipes. Therefore, in a number of applications (it is especial in the heating engineer) sometimes it is necessary to carry out special processing of water with the purpose of achievement of an optimum ratio between rigidity of water and its corrosion activity. EXPERIMENTAL PART The adsorption processes including both processes cleanly including physical, and the processes conducting to formation of new chemical substances are rather various. Processes concern to them ion-exchange chromatographies proceeding between ionits and solutions of electrolit also. Due to the universality and these processes have found of flexibility wide application in analytical chemistry, food and an iron and steel industry, in power system, water-preparation and in many other areas of a science and technics(technical equipment), and now supersede from an industrial practice other methods sorption [1-3]. Despite of the big assortment industrial ionits, the majority of them, especially, condensation type differ such low parameters, as low speed sorption, small stability to thermal, chemical influences, etc. It limits opportunities and spheres of their application [4,5]. By us it is received new monofunctional sulphocationite on the basis of interaction of styrene with an accessible and cheap waste of agricultural and cotton scraping industries. SHAPE * MERGEFORMAT
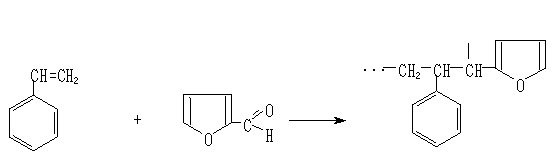 Sorption abilities of sulphocationite to ions of calcium, magnesium, copper, nickel carried out research from 0.1N solutions of СаСl2, MgCl2, CuSO4 and NiSO4. Sorption of calcium and magnesium determined by trilonometric, copper by iodometric, nickel by photocolorimetric methods [10,11]. According to the literary data, polymers and ion-exchangers, received on the basis of derivatives of furan differ from raised thermal and chemical stability and mechanical durability [5,6]. The basic properties of received sulphocationite are submitted in table 1. Table 1 The main physical and chemical parameters of received sulphocationite
As is known in many regions of our republic used in a life and on manufacture water has high rigidity which sometimes reaches up to 12 mg-ecv/l instead of 2874-82 "Potable water" received according to GOST – 2.5-7 mg-ecv/l. From the data of table 1 it is visible, that the examinee cationit possesses high enough parameters of size of exchange capacity on ions of calcium and magnesium. As object of research we had been used artesian water of area Shurchi in Surkhan-Darya area which has rigidity of 12.2 mg-ecv/l. Cationits tested in Н-and Na-forms. 1 gr. of cationit filled in 200 ml of researched water. In 24 hours water separated from cationit and determined its rigidity trilonometric method at the presence of the indicator chromogen black. Thus rigidity of water in the Н-form has made 4.5 mg - ecv/l, and in the Na-form of 2.2 mg - ecv/l. Except for it we had been carried out researches on softening water of 2.5 mg-ecv/l having rigidity. After contact within 6 hours of water with cationit rigidity of it corresponded at use cationit in: the Н-form – 0.8 mg-ecv/l; the Na-form – 0.4 mg-ecv/l. Results of the received researches testifies to perspectivity of work with the examinee with sulphocationite. Vocabulary
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Просмотров: 527 | Загрузок: 0 | | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Всего комментариев: 0 | |